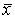 |
关键词:必降脂;游离脂肪酸;胰岛素敏感指数
【摘要】 目的 为探讨降脂药物――必降脂对糖代谢的影响。方法 将伴有高脂血症的74例2型糖尿病患者随机分成2组,保持原糖尿病治疗方案不变。治疗组每天加服必降脂缓释片400mg,对照组不加服。结果 3个月以后发现治疗组血胆固醇、甘油三酯,LDL-C,空腹血糖有较明显下降,HDL-C轻度上升,糖耐量改善,胰岛素敏感指数上升。而对照组无显著变化。结论 必降脂除了能改善脂代谢紊乱以外还有助于纠正糖代谢异常。
Effect of antihyperlipidemic drug-bezafibrate on glucose metabolism Yang Yugou,Lin Dongping,Sheng Hongguang,et al.The Department of Internal Medicine,Ninth People's Hospital, Shanghai Second Medical University,Shanghai,200011
【Abstract】Objective Tostudytheeffectofantihyperlipidemicdrug-bezafibrate on glucose metabolism. Methods 74 cases of type 2 diabetes mellitus patients associated with hyperlipidemia were randomly divided into 2 groups. The original antihyperglycemic therapies were maintained. The treatment group received bezafibrate sustained-release tablet 400mg/d,thecontrolgroupdidnotreceiveadditional drug. Results After three months, in the treatment group serum cholesterol, triglyceride, low-density lipoprotein cholesterol and fasting glucose decreased obviously, high-density lipoprotein cholesterol increased slightly, glucose tolerance improved and insulin sensitivity index increased. These changes were not observed in the control group. Conclusion In addition to improve lipidemic disturbance, bezafibrate had the additional effect of correcting glucose metabolism disturbance.
【Key words】 Bezafibrate Free fatty acid Insulin sensitivity index
(Chin J Endocrinol Metab, 1998, 14:103-105)
我们对有脂代谢紊乱的2型糖尿病患者用必降脂缓释片(每片含苯扎贝特400mg)治疗时可见该药在使血胆固醇(TC)、甘油三酯(TG)和游离脂肪酸(FFA)下降时也使空腹血糖下降、糖耐量改善,提高了患者的胰岛素敏感度。
对象和方法
一、方法
74例伴有脂代谢紊乱的2型糖尿病患者。病程3~17年(12±4年)。开始进入试验时糖化血红蛋白浓度为8.55%~14.4%(9.65±0.5%),正常值<7.5%。甘油三酯(TG)2.7±0.5 mmol/L,总胆固醇(TC)6.6±0.2mmol/L,肝,肾功能正常。均以饮食控制加磺脲类降糖药(格列齐特)治疗。参试前分别测定空腹血糖、HbA1 、TG、TC、LDL-C、HDL-C、尿酸、BUN、Cr和肝功能。停用一切降脂药1月以后再次测定TG、TC、LDL-C、HDL-C和空腹血糖。作OGTT(口服75克葡萄糖法),同时测定各点的血胰岛素和FFA值。然后随机抽样法分成治疗组(每人每天加服必降脂缓释片400mg共3月)和对照组(不服必降脂但仍维持原糖尿病治疗方案)各37人。服用必降脂后1月、2月、3月分别测定TG、TC、LDL-C和HDL-C,试验结束时复查HbA1 、尿酸、BUN、Cr和肝功能,重复OGTT并测定各点的胰岛素和FFA值计算出患者的胰岛素敏感指数〔1〕 。
二、统计方法
用均值±标准差(±s)显示。用t检验进行比较,对治疗前后空腹血糖值和FFA值之间的变量关系用相关分析法处理。并对相关系数r作t检验。
结 果
一、临床资料以及对照组和治疗组的空腹血糖值、HbA1 值、体重指数、年龄各项间无统计学差异,P>0.05,见表1。
二、血脂变化见表2,停用其他降脂药物1月前后治疗组和对照组血脂无显著差异,P>0.05。必降脂治疗3月以后血脂水平明显改善。而对照组试验前后无显著差异,P>0.05。
三、血糖、胰岛素和FFA变化见表3。治疗组治疗前糖耐量曲线下面积为2406±96.2mmol/min,治疗后下降到2166±86.6mmol/min,P<0.05。对照组变化不明显。胰岛素曲线下面积治疗组从5730±120.4mU/min下降到4566±86.6mU/
表1 开始试验时的临床资料(±s)
Tab 1
Clinical data before treatment (±s)| 治疗组 | 对照组 | |
| Treatment group | Control group | |
| 例数(男/女) | 17/20 | 18/19 |
| Number(M/F) | ||
| 年龄(岁) | 54.7±1.8 | 51.6±2.6 |
| Age(yr) | ||
| 体重指数(kg/m2 ) | 26.6±0.8 | 25.9±0.9 |
| Body mass index (kg/m2 ) | ||
| 空腹血糖(mM) | 11.6±0.8 | 9.8±0.9 |
| FBG(mmol/L) | ||
| HbA1 (%) | 10.1±0.3 | 9.1±0.7 |
表2
必降脂对血脂的影响(单位:mmol/L)Tab 2
Effect of bezafibrate on serum lipid concentrations(mmol/L)| 开始试验时 均值(±s) At beginning of study (±s) | 停用其它降脂药 一月以后(±s) One month after withdrawl of other antihyperlipidemic agents(±s) | 必降脂治疗后的变化(±s) The changes after bezafibrate | ||||
| 1月 1 mon | 2月 2 mon | 3月 3 mon | ||||
| TG | 治疗组 Treatment group | 2.7±0.5 | 2.2±0.2 | 1.7±0.2* | 1.7±0.2* | 1.4±0.2** |
| 对照组 Control group | 2.4±0.3 | 1.8±0.2 | 2.1±0.2 | 2.1±0.2 | 1.9±0.2 | |
| TC | 治疗组 Treatment group | 6.6±0.2 | 6.3±0.3 | 5.6±0.3 | 5.6±0.2* | 5.5±0.2** |
| 对照组 Control group | 6.4±0.2 | 6.2±0.2 | 6.4±0.2 | 6.4±0.3 | 6.4±0.2 | |
| LDL-C | 治疗组 Treatment group | 4.2±0.3 | 3.6±0.3 | 3.6±0.3 | 3.7±0.2 | 3.5±0.2* |
| 对照组 Control group | 4.3±0.2 | 4.2±0.2 | 4.2±0.2 | 4.2±0.3 | 4.1±0.3 | |
| HDL-C | 治疗组 Treatment group | 1.2±0.1 | 1.2±0.1 | 1.3±0.1 | 1.3±0.1 | 1.3±0.1 |
| 对照组 Control group | 1.1±0.1 | 1.2±0.1 | 1.2±0.1 | 1.3±0.1 | 1.3±0.1 | |
表3
必降脂对糖代谢的影响(±s)Tab 3
Effect of bozafibrate on glucose metabolism(±s)| 空腹 Fasting | 30' | 60' | 120' | 180' | |||||||
| 治疗组 Treatment | 对照组 Control | 治疗组 Treatment | 对照组 Control | 治疗组 Treatment | 对照组 Control | 治疗组 Treatment | 对照组 Control | 治疗组 Treatment | 对照组 Control | ||
| 血糖 Glucose (mmol/L) | 治疗前 before | 10.6±0.6 | 9.8±0.5 | 12.5±0.5 | 12.1±0.7 | 15.2±0.7 | 14.2±0.6 | 14.0±0.5 | 13.3±0.5 | 11.6±0.6 | 11.2±0.4 |
| 治疗后 after | 9.1±0.7** | 9.88±0.3 | 12.1±0.4 | 12.5±0.4 | 14.1±0.7* | 13.6±0.4 | 12.1±0.6** | 13.8±0.5 | 10.2±0.5* | 10.3±0.5 | |
| 胰岛素 Insulin (mU/L) | 治疗前 before | 10.1±0.5 | 10.2±0.3 | 35±2 | 34±2 | 40±2 | 39.5±1 | 30.9±0.9 | 20.0±1 | ||
| 治疗后 after | 7.2±0.1* | 10.2±0.2 | 33±1.5* | 35±3 | 29±1** | 39±2 | 21±0.5* | 21±0.5 | |||
| FFA (mmol/L) | 治疗前 before | 0.64±0.06 | 0.62±0.05 | 0.43±0.05 | 0.37±0.05 | 0.34±0.06 | 0.32±0.02 | 0.23±0.04 | 0.22±0.01 | 0.2±0.04 | 0.14±0.01 |
| 治疗后 after | 0.42±0.04* | 0.58±0.05 | 0.33±0.04* | 0.36±0.03 | 0.21±0.02* | 0.37±0.02 | 0.14±0.01** | 0.19±0.01 | 0.15±0.02** | > Transfer interrupted! | |